Lawrenceville, NJ (February 18, 2004) - The United States Patent and Trademark Office has issued a patent to Vela Pharmaceuticals Inc. (www.velapharm.com) for the use of tianeptine in irritable bowel syndrome (IBS) and nonulcer dyspepsia (NUD). Vela Pharmaceuticals (VelaPharm) is a privately held pharmaceutical company specializing in the “rediscovery” and development of neurotherapeutic medicines. Tianeptine is currently marketed outside the United States for the treatment of depression. Vela Pharmaceuticals Awarded Patent for use of Tianeptine in Irritable Bowel Syndrome (IBS) and Nonulcer Dyspepsia (NUD)
Company Plans Phase 2 Trial in Both Women and Men with IBSTianeptine´s mechanism of action appears to be different from that of other marketed antidepressants. In contrast to the selective serotonin reuptake inhibitors (SSRIs), such as Prozac® and Paxil®, tianeptine enhances the reuptake of serotonin. Serotonin plays a key role not only in the brain, but also in the enteric nervous system (ENS) within the intestinal tract, where it mediates peristaltic and secretory reflexes. Serotonergic overactivity within the intestines increases these reflexes and thereby contributes to the pain, bloating, and diarrhea suffered by IBS patients. Tianeptine may attenuate IBS symptoms by decreasing serotonergic activity within the gut.
US Patent No. 6,683,072 was issued to VelaPharm on January 27, 2004. VelaPharm filed the patent application in early 2003 after discovering that tianeptine was active in an animal model for IBS. According to Robert Kucharik, Director of Pharmacology at VelaPharm, “The results of our preclinical effort provided a signal for potential efficacy in functional gastrointestinal disorders such as IBS.”
IBS is the disorder most commonly diagnosed by gastroenterologists in the United States. It affects the lives of 15 percent of US adults and accounts for 12 percent of visits to primary care physicians, costing more than $8 billion each year in direct medical expenses. Yet most current treatments have limited application, carry substantial risks, and focus on symptomatic relief.
VelaPharm, which already has one asset (dextofisopam) in a Phase 2 IBS trial, plans to initiate a separate Phase 2 clinical trial later this year to test tianeptine´s usefulness in treating IBS. “Tianeptine has been prescribed outside the United States for the treatment of depression since the late 1980s,” says Steven M. Leventer, PhD, Senior Vice President of VelaPharm´s Research and Development department. “We believe that our work with tianeptine may eventually help to relieve the pain and social disability that IBS inflicts on millions of women and men worldwide.”
About VelaPharm
Vela Pharmaceuticals Inc. (www.velapharm.com), a privately held company, specializes in the “rediscovery” and development of medicines to treat diseases associated with the nervous system. The company uses its extensive drug development expertise to leverage the value of “known” neurotherapeutic molecular assets. Investors include JP Morgan Partners (www.jpmorganpartners.com), New Enterprise Associates (www.nea.com), and Venrock (www.venrock.com).
* * * [NOTE: On 15 March 2006, Pharmos Corporation (Nasdaq: PARS) "announced today that it has entered into a definitive agreement to acquire Vela Pharmaceuticals Inc., a venture-capital backed, privately owned company specializing in the development of medicines related to diseases of the nervous system including disorders of the "brain-gut axis."]
Refs
further reading about tianeptineHOME
Search
Amineptine.com
Servier Laboratories
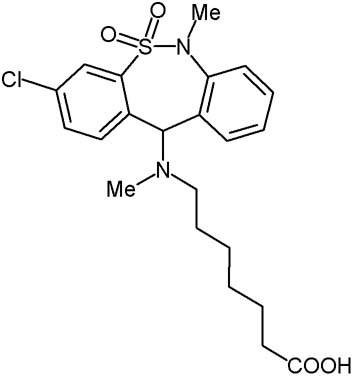
Tianeptine: structure
THE GOOD DRUG GUIDE
International Pharmacies 2006
Tianeptine (Stablon): product info from manufacturer
c o n t a c t
dave@bltc.com